Fully automated, walk-away, random access instrument for the Immuno Fluorecence Assay on Glass.
UNIQO is the new high-performance automated system for the IFA procedure, from slide preparation to test reading and validation. It provides laboratories with an all-in-one solution that integrates automated IFA protocol with complete slide mounting, including coverslip, image acquisition, analysis and archiving.
UNIQO supports standardization by eliminating the inconsistency of human intervention and maximizing the accuracy of the overall IFA process, from accurate dispensing and mounting of IFA slides, to more objective image acquisition and analysis.
Innovative software manages and controls the entire analytical workflow ensuring optimal slide processing and image acquisition for fully automated analysis of immunofluorescence signals and effective data management.
In addition, the integrated virtual microscope allows navigation and review of the digitized slide from remote locations, eliminating the problem of storing the slides and preserving the fluorescent signal over time. The Indirect Fluorescence Immune Expert can focus directly on the results and reporting.

Features
- Automated IIF slide preparation
- Automated mounting of the coverslip
- Preservation of slides
- Walkaway process
- Continuous access
- Complete scan of wells
- Automatic image acquisition of IIF tests
- Automatic determination of the positive/negative sample
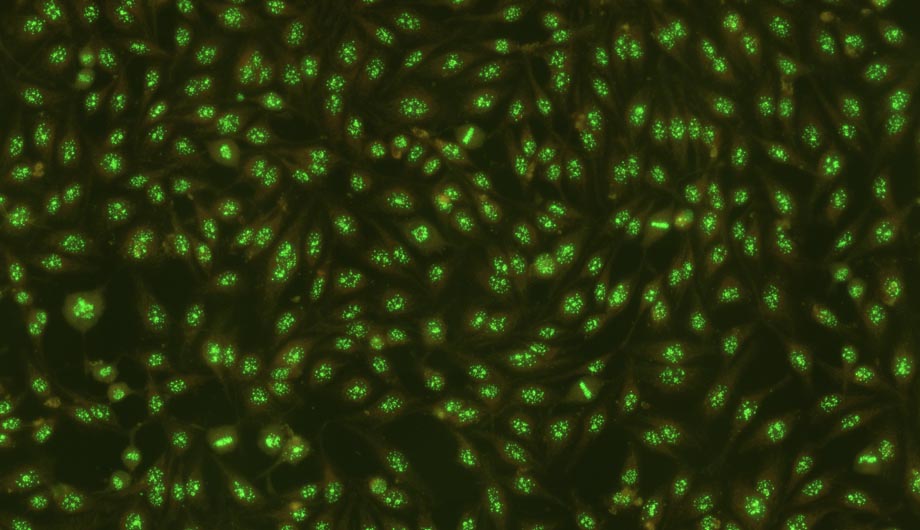
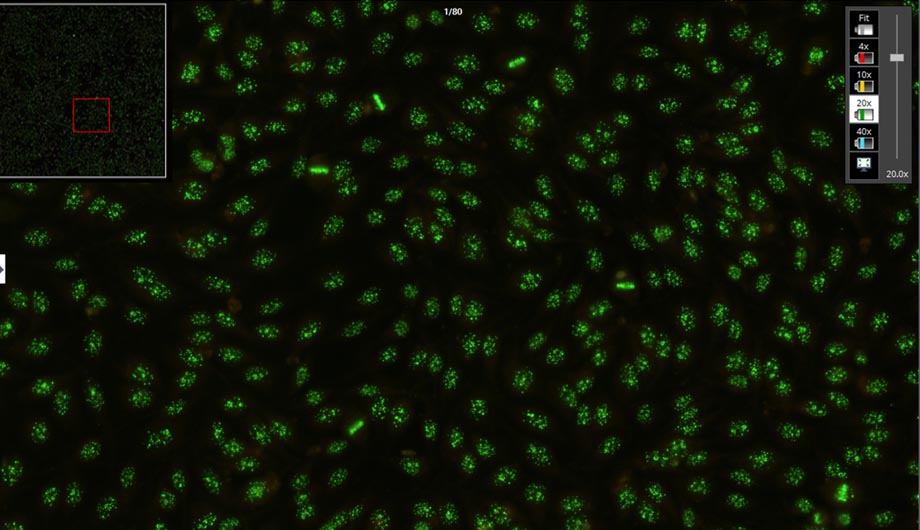
- HEp-2 pattern recognition
- Digitization of other IIF substrates
- Automatic determination of titration
- Archiving of digital results
- Virtual microscope
- Connection to LIS/middleware
- Full data traceability (1D/2D barcode reading)
- On-board PC and touch-screen monitor
UNIQO

- Up to 18 slides per run per workstation
- Up to 160 patients per run per workstation
- Up to 264 predilution cuvettes
- Image acquisition speed: about 30 seconds/well
- Cost and time savings
- Up to 18 slides per run per workstation
- Up to 160 patients per run per workstation
- Up to 264 predilution cuvettes
- Image acquisition speed: about 30 seconds/well
- Cost and time savings
- Automatic mounting of the coverslip without imperfections
- Lids are automatically placed to preserve slides throughout the procedure
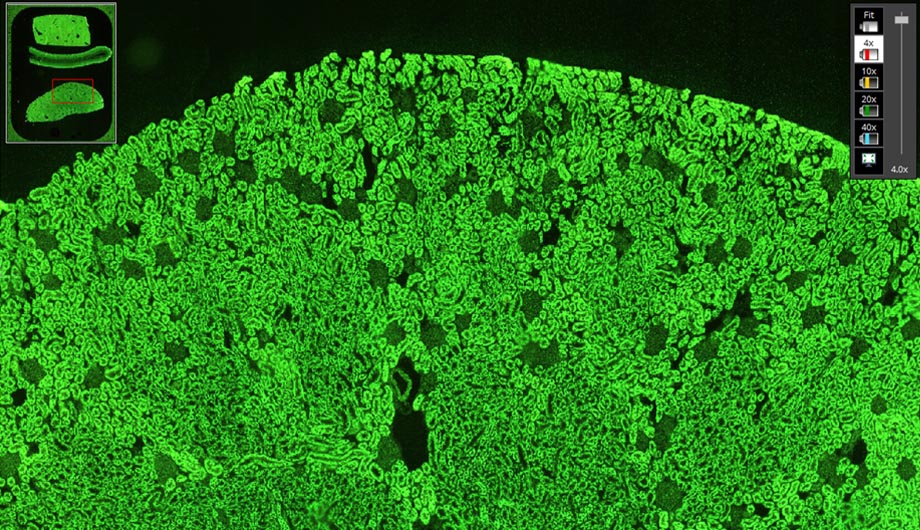
- Slide digitization: excellent image acquisition with high emissions even at low magnifications
- Fully traceable processing (barcode reading of samples and slides)
- Complete digitization and archiving
- Virtual microscope for remote retrieval, review and second opinion
- Walkaway process with continuous access
- Automatic determination of titration
Customers rely on us for quality, experience in manufacturing IVD medical devices and our capabilities in developing new products. And they talk about it, worldwide.
A Fully Automated Platform Capable of Processing, Scanning and Analyzing IFA Substrates
John Breneman, Sarah Paul, Jeri Seiki, Roger Walker
Clinical Immunology Division, Bio-Rad Laboratories, 4000 Alfred Nobel Drive, Hercules, California 94510.
Presented at the 13th Dresden Symposium on Autoantibodies – Dresden, Germany – September 27 – 30, 2017
IFA Substrate Processing, Scanning and Analysis on the IFx 360 System, a Fully Automated Platform
John Breneman, Sarah Paul, Jeri Seiki, Cindy Liedstrand
Clinical Immunology Division, Bio-Rad Laboratories, 4000 Alfred Nobel Drive, Hercules, California 94547
ZENIT-PRO, A FULLY AUTOMATED INDIRECT IMMUNOFLUORESCENCE ANALYSER
A preliminary evaluation of the analytical performance
Martina Fabris, Daria Franceschi, Daria Picchioni, Elio Tonutti, Nicola Bizzaro
On behalf of the Study Group on Autoimmune Diseases of the Italian Society of Laboratory Medicine, Italy – Institute of Clinical Pathology and Immunopathology and Allergy, University Hospital of Udine, Italy – A. Menarini Diagnostics, Firenze, Italy – VISIA Imaging Srl, San Giovanni Valdarno, Firenze, Italy – Chairman, Study Group on Autoimmune Diseases, SIPMeL, Italy
IMMUNOLOGY
Next-generation IFA testing: teaching an old dog new tricks
Jake Morrow
www.pathologyinpractice.com
October 2018


